Case Study Strengthening and Stabilizing Diagnostic Laboratory Operations--Lessons I learnt
By Dr. Arpan Gandhi
drarpangandhi.org and arpangandhi@gmail.com
Context
In one engagement, I worked with a diagnostic laboratory that was already functional, accredited, and processing a steady volume of samples. On paper, it appeared stable. Systems existed, documentation was in place, and the team was experienced.
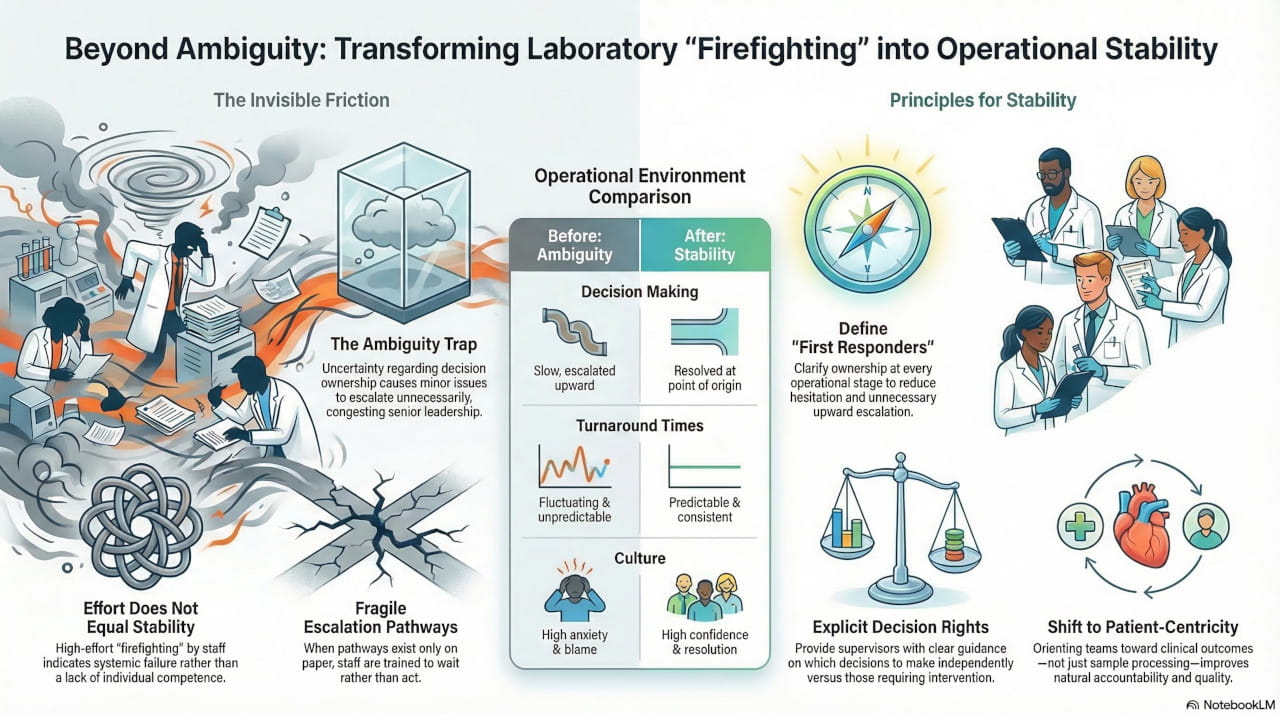
However, once I began observing day-to-day operations more closely, a different picture emerged. The laboratory was constantly firefighting. Turnaround times fluctuated unpredictably. Supervisors felt stretched and reactive. Senior leadership remained involved in operational decisions that should not have required their attention.
Everyone was working hard. Yet the system felt fragile.
From experience, I have learned that when effort is high but stability is low, the issue is rarely individual competence. It is almost always systemic.
My Initial Observations
Rather than starting with SOPs, audit reports, or dashboards, I focused on understanding how work actually flowed through the laboratory.
What became clear early on was that roles were broadly defined but weakly owned. Decision-making authority was unclear at several points. Escalation pathways existed on paper but were inconsistently followed in practice. Quality issues were addressed reactively, often after they had already created stress.
Supervisors escalated issues upward not because they lacked capability, but because they lacked clarity and confidence in their authority. This created congestion at senior levels and delayed resolution. Over time, the system trained people to wait rather than act.
Framing the Core Problem
In my experience, operational instability rarely arises from complexity. More often, it arises from ambiguity.
When people are uncertain about who owns a decision, work either stalls or escalates unnecessarily. In this laboratory, many issues that should have been resolved locally were moving upward simply because decision boundaries were unclear.
My focus was therefore not on adding new controls, but on reducing uncertainty within the existing structure.
My Approach
I approached this engagement with three guiding principles:
- Simplify before adding
- Clarify ownership at every operational stage
- Support people rather than supervise them more closely
I spent time mapping workflows as they were actually practiced, not as they were documented. Particular attention was paid to pre-analytical handling, reporting bottlenecks, and quality-related escalations, as these areas generated the most daily stress.
Interventions Implemented
The interventions were deliberately practical and incremental.
We first clarified ownership at each operational stage so that every process had a clearly defined first responder. This reduced hesitation and unnecessary escalation.
Next, we defined decision rights for supervisors and section heads. This included explicit guidance on what they were expected to decide independently and what truly required senior involvement.
Escalation pathways were then simplified into predictable, time-bound structures. Instead of multiple informal routes, issues followed a clear and consistent path.
Quality monitoring was aligned with real risk points rather than broad indicators, allowing teams to focus attention where it mattered most. Finally, we reduced dependence on individuals by standardising routine decisions, ensuring continuity even during absences or transitions.
A Personal Turning Point
One moment that stayed with me was realizing how disconnected some team members felt from patient outcomes. Because they did not see patients daily, there was a tendency to view their work as routine test running and analysis. I had to consciously orient the team to understand that every step they performed had a direct impact on patient management. Shifting this mindset—from processing samples to influencing clinical decisions—changed how people approached quality and responsibility.
What Changed Over Time
As systems stabilised, several shifts became evident. Operational noise reduced significantly. Supervisors became more confident in decision-making. Issues were resolved closer to their point of origin. Senior leadership was able to step back from daily firefighting and focus on oversight rather than intervention.
Importantly, these improvements did not come from increased workload or tighter monitoring. They emerged because ambiguity had been reduced.
Turnaround times became more predictable. Quality discussions became more meaningful. Teams reported feeling more in control of their work.
Impact on People and Culture
One of the most encouraging changes was cultural. As clarity improved, anxiety reduced. People stopped second-guessing themselves. Conversations shifted from blame to resolution.
When systems support people, confidence follows. When confidence improves, quality improves naturally.
This reinforced a belief I have seen repeatedly: culture improves when systems become fair, predictable, and transparent.
Key Learning
This engagement reinforced a principle I now apply consistently:
Most laboratories do not need more policies, more technology, or more oversight. They need systems that allow competent people to function without constant escalation.
Stability is not achieved by tightening control. It is achieved by reducing ambiguity.
Reflection
Running a diagnostic laboratory well is not about perfection. It is about creating systems that can absorb pressure without transmitting stress downward.
When operations are stabilised, quality improves as a consequence—not as a parallel initiative.
Why This Matters
This approach allows laboratories to:
- Improve reliability without disruption
- Reduce burnout at supervisory levels
- Strengthen quality without increasing audit fatigue
- Build confidence and accountability across teams
In my experience, calm systems consistently produce better outcomes than busy ones